- Visibility 347 Views
- Downloads 49 Downloads
- DOI 10.18231/j.jpbs.2024.017
-
CrossMark
- Citation
AI in autoimmune diseases: Transforming diagnosis and treatment
Introduction
The large and complicated category of conditions known as autoimmune diseases is defined by an aberrant immune response directed against the body's own tissues. Protecting the body from foreign invaders like bacteria, viruses, and other diseases is the main job of the immune system in a healthy person. It accomplishes this by differentiating between self and non-self-antigens, a function that depends on a finely synchronized cell and signaling molecule network. This self-recognition process malfunctions in people with autoimmune illnesses, though, and the immune system unintentionally targets its own cells, tissues, or organs. Chronic inflammation, tissue damage, and reduced organ function are all possible outcomes of this abnormal immune response, which can lead to serious morbidity and, in extreme situations, death.[1], [2]
Overview and Classification of Autoimmune Diseases
Organ-specific and systemic autoimmune illnesses are the two primary types of autoimmune diseases. Autoimmune illnesses that target a particular organ or tissue are known as organ-specific disorders. For instance, Hashimoto's thyroiditis and Graves' disease target the thyroid gland, causing hypothyroidism and hyperthyroidism, respectively, whereas Type 1 diabetes mellitus (T1DM) typically affects the pancreatic beta cells, resulting in insulin shortage and hyperglycemia. Systemic autoimmune disorders, on the other hand, impact several organs or systems at once. Systemic autoimmune illnesses include conditions like systemic lupus erythematosus (SLE), rheumatoid arthritis (RA), and systemic sclerosis (SSc), in which the immune response is more extensive and affects multiple tissues and organs, such as the skin, joints, kidneys, and lungs.[3], [4], [5]
Epidemiology and Burden of Autoimmune Diseases
Approximately 5% to 10% of people worldwide suffer from autoimmune illnesses, which are more common in women than in men. A mix of environmental, hormonal, and genetic factors are responsible for this gender gap. In addition to the high expense of medical care, autoimmune illnesses have a major negative influence on the quality of life for those who are afflicted. Chronic pain, exhaustion, and disability are common among patients, and they can cause social isolation, psychological discomfort, and a decline in productivity. Furthermore, managing autoimmune disorders frequently takes a lifetime, placing a constant financial burden on both patients and healthcare systems.[6], [7]
Pathogenesis of Autoimmune Diseases
Environmental factors, immune system dysregulation, and genetic predisposition all play intricate roles in the multifactorial pathophysiology of autoimmune disorders. Susceptibility to autoimmune disorders is significantly influenced by genetic factors. A higher risk of autoimmune diseases has been closely linked to specific genetic loci, especially those found in the major histocompatibility complex (MHC). For example, certain human leukocyte antigen (HLA) alleles have been linked to the pathophysiology of certain diseases, such as HLA-DR4 in rheumatoid arthritis and HLA-DR3/DR4 in Type 1 diabetes. In addition to HLA, other immune-regulating genes like CTLA-4, PTPN22, and IL-23R have also been connected to a number of autoimmune disorders. In people who are genetically predisposed, environmental stimuli such as stress, chemicals, illnesses, and food items are thought to serve as triggers.[8], [9], [10], [11], [12] For instance, autoimmune illnesses like multiple sclerosis (MS) and systemic lupus erythematosus (SLE) have been linked to viral infections like Epstein-Barr virus (EBV) and cytomegalovirus (CMV). Cross-reactivity and autoimmunity can also result via molecular mimicry, in which foreign antigens and self-antigens have structural similarities. Autoimmune illnesses are mostly caused by dysfunction of the immune system. Autoimmunity is characterized by a loss of both central and peripheral self-tolerance. Through negative selection, central tolerance mechanisms in the bone marrow and thymus eradicate self-reactive T and B cells. It is possible for certain autoreactive lymphocytes to evade deletion and make their way into the peripheral circulation. These escaping cells are managed by immunological checkpoints including PD-1 and CTLA-4, anergy, and peripheral tolerance mechanisms like regulatory T cells (Tregs). These tolerance systems malfunction in autoimmune disorders, enabling autoreactive cells to multiply and launch an immunological attack on self-tissues.[13], [14], [15]
Discussion
Current treatment strategies and limitations
The main goals of managing autoimmune illnesses are to reduce the aberrant immune response, lower inflammation, and control symptoms. Nonsteroidal anti-inflammatory drugs (NSAIDs), corticosteroids, disease-modifying antirheumatic medications (DMARDs), and biologic agents that target particular immune system components—such as tumor necrosis factor (TNF) inhibitors and interleukin-6 (IL-6) receptor antagonists—are examples of current treatment approaches. These treatments have drawbacks even if they may be useful in controlling illness symptoms and slowing the course of the condition. Significant adverse effects are linked to several of these medicines, such as heightened vulnerability to infections, gastrointestinal problems, and, in the case of corticosteroids, long-term consequences like osteoporosis and adrenal suppression. Furthermore, because these treatments don't address the root cause of the autoimmune reaction, they frequently need to be administered for the rest of one's life. This can result in drug resistance and necessitate combination therapy or drug change.[16], [17], [18]
The Role of Artificial Intelligence in Autoimmune Diseases
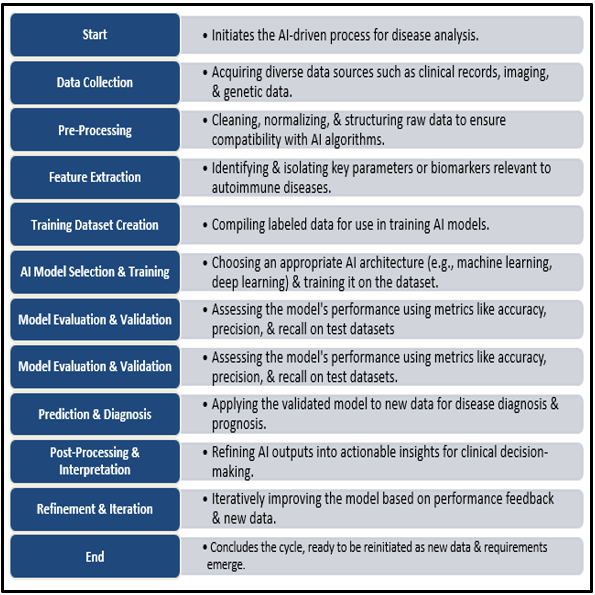
The detection and treatment of autoimmune illnesses could be revolutionized by recent developments in artificial intelligence (AI). Large and complex datasets, such as genetic, proteomic, and clinical data, may be analyzed by AI technologies, especially machine learning (ML) and deep learning (DL), which could lead to the discovery of new biomarkers and more accurate predictions of disease risk, onset, and progression than conventional techniques. AI can improve image analysis in diagnostics, helping to identify minute tissue alterations that might be a sign of autoimmune disease activity. Additionally, by combining patient-specific information, such as genetic and molecular profiles, AI can support individualized treatment strategies by customizing medications to optimize effectiveness and reduce negative effects. As the flow of AI's function in managing autoimmune diseases shown in ([Figure 1]).[19], [20], [21]
Ai in diagnosis and biomarker discovery
Medicine is changing quickly due to artificial intelligence (AI), especially in the diagnosis and treatment of complicated illnesses like autoimmune diseases. Early diagnosis of autoimmune disorders is severely hampered by their variety and overlapping clinical manifestations, which frequently results in inappropriate or delayed treatments. The limitations of conventional diagnostic approaches, which mostly rely on clinical examination and standard laboratory testing, include their incapacity to accurately forecast the development of disease or identify subtle changes in disease patterns. In this regard, artificial intelligence (AI) technologies—specifically, machine learning (ML) and deep learning (DL) algorithms—offer a revolutionary way to enhance the accuracy of autoimmune disease diagnosis and the identification of biomarkers. AI systems have proven to be remarkably adept at processing large volumes of diverse data, including imaging data, clinical records, and multi-omics datasets like proteomics, metabolomics, and genomes.[22], [23] AI can find patterns and correlations in these intricate datasets that are difficult for human therapists or conventional statistical techniques to detect. AI algorithms can combine many kinds of data to create predictive models that improve the accuracy of autoimmune disease detection. For example, based on minor variations in clinical, serological, and genetic data, machine learning models can be trained on sizable patient data sets to distinguish between autoimmune conditions that exhibit comparable symptoms, such as rheumatoid arthritis and systemic lupus erythematosus. This may improve patient outcomes by enabling prompt action and drastically cutting down on diagnostic delays.[24], [25], [26]
In addition to increasing diagnostic precision, AI is essential for finding biomarkers for autoimmune disorders. For the diagnosis, prognosis, and tracking of treatment response, biomarkers are essential. However, because of the intricate pathophysiology and wide range of disease manifestations, it has been difficult to find trustworthy biomarkers for autoimmune diseases. By examining high-dimensional datasets, AI-driven methods—in particular, DL algorithms—have demonstrated potential in identifying new biomarkers. For instance, DL algorithms can examine patient gene expression data to find distinct molecular signatures linked to particular autoimmune disorders. Through the identification of patients who are more likely to respond to particular medicines, these AI-derived biomarkers can not only help with early diagnosis but also enable individualized treatment plans. The Biomarkers Identified Using AI for Autoimmune Diseases mentioned in ([Table 2]).[27], [28], [29]
Through wearable technology and sophisticated imaging methods, AI technologies also make it possible to analyze and continuously track the course of diseases in real time. AI-powered imaging analysis can identify subtle tissue alterations that indicate autoimmune disease activity or remission. AI systems, for example, can examine radiographs of joints in rheumatoid arthritis to find early indications of inflammation or erosion before clinical symptoms appear. In a similar vein, wearable biosensors that continually measure physiological parameters can be combined with AI to provide real-time input on disease activity and enable more flexible and rapid treatment modifications. AI has enormous promise to revolutionize autoimmune disease diagnosis and biomarker development, but there are still a number of obstacles to overcome. The requirement for high-quality, labeled data in order to effectively train AI algorithms is one of the main issues. Biases in training datasets and variations in data quality can result in errors and restrict how broadly AI models can be applied to various populations. Furthermore, to provide fair access to AI-driven diagnostic tools, ethical issues pertaining to data privacy, security, and the possibility of algorithmic bias must be properly controlled.[30], [31], [32], [33], [34]
Going forward, thorough validation of AI models in a variety of patient groups and real-world contexts is necessary for the incorporation of AI in clinical practice for autoimmune illnesses. To provide uniform standards for the creation and application of AI technology, cooperation between data scientists, physicians, and regulatory agencies is crucial. The field can progress toward more precise, early diagnosis and individualized treatment of autoimmune illnesses by tackling these issues and utilizing AI's advantages, which will ultimately improve patient outcomes and lower healthcare expenses. AI has enormous potential to improve autoimmune disease biomarker identification and diagnostic precision, providing a route to more individualized and accurate medical treatment. AI technologies will surely become more and more important in changing the way autoimmune diseases are managed as they develop and become more integrated into clinical practice. The Comparison of AI Models in Autoimmune Disease Management are mentioned in ([Table 1]).[35], [36]
Personalized treatment approaches
Considering the variety and complexity of autoimmune disorders, personalized therapy options have become a viable approach. The necessity for specialized treatment approaches is highlighted by the wide range of clinical symptoms and inconsistent responses to treatment that characterize autoimmune disorders like multiple sclerosis, lupus, and rheumatoid arthritis. Individual differences in illness pathophysiology, genetic background, and environmental factors that affect patient outcomes may not be taken into consideration by traditional treatment paradigms, which frequently use a one-size-fits-all approach. Many patients suffer from poor therapeutic outcomes, negative drug reactions, or illness recurrence as a result. In this regard, artificial intelligence (AI) provides a potent instrument to improve individualized treatment strategies by combining multifaceted data to forecast treatment reactions, optimize treatment plans, and eventually boost patient outcomes. The AI-Driven Personalized Treatment Examples are mentioned in ([Table 3]).[37], [38], [39]
In order to examine huge and complex datasets, such as genomic, transcriptomic, proteomic, and clinical data, AI-driven tailored treatment for autoimmune diseases makes use of machine learning (ML) and deep learning (DL) methods. AI can more accurately forecast how each patient will react to particular treatments by finding patterns and connections that human clinicians are unable to see. For example, ML algorithms can stratify patients according to their propensity to respond to a specific biologic therapy, such as interleukin-6 (IL-6) antagonists or tumor necrosis factor (TNF) inhibitors, by analyzing patient-specific data, such as immune cell signatures, gene expression profiles, and genetic polymorphisms. By using this stratification, doctors may choose the best course of action for every patient, minimizing trial-and-error methods and lowering the possibility of side effects from unsuccessful treatments.[40], [41] AI can help determine the best dosage and timing of treatments in addition to optimizing drug selection, which will further improve individualized treatment plans. Subtherapeutic or hazardous medication levels might result from traditional dosing regimens' frequent failure to take into consideration inter-individual heterogeneity in drug metabolism, distribution, and clearance. In order to customize dose plans, AI algorithms can incorporate pharmacokinetic and pharmacodynamic data in addition to patient-specific factors including age, weight, comorbidities, and concurrent drugs. AI, for instance, can forecast the ideal medication dosage and timing for autoimmune conditions that call for immunosuppressive therapy in order to enhance effectiveness while lowering the risk of infections and other side effects. This improves patient safety and treatment compliance.[42], [43], [44]
AI also makes it easier to integrate real-time patient data from electronic health records (EHRs), wearable technology, and remote monitoring tools so that treatment plans can be dynamically modified in response to patient feedback and continuing disease activity. A more sensitive and adaptable approach to treatment is made possible by this ongoing monitoring, where AI algorithms examine data streams to identify early indicators of a disease flare or remission and modify treatments accordingly. AI-powered monitoring, for instance, can allow for prompt modifications to treatment plans in conditions like multiple sclerosis, where disease activity can fluctuate greatly over time. This could potentially avert permanent harm and improve long-term results. Furthermore, AI-driven tailored therapy strategies for autoimmune disorders go beyond medication to include nutritional advice, lifestyle changes, and psychosocial support—all essential elements of all-encompassing disease management. AI can offer personalized recommendations that support pharmacological treatments and address the complex nature of autoimmune disorders by evaluating patient-specific data, such as stress levels, nutritional status, and gut microbiota composition. In addition to increasing treatment effectiveness, this all-encompassing strategy gives patients the confidence to actively manage their illness, which raises adherence and improves quality of life. [45], [46], [47]
Even though AI holds promise for improving individualized care for autoimmune disorders, a number of obstacles need to be overcome before its full potential can be achieved. High-quality, consistent data and thorough testing of AI algorithms across a range of patient groups are necessary for the integration of AI into clinical practice in order to guarantee the accuracy and generalizability of the algorithms. To foster trust between patients and healthcare practitioners, ethical issues including data protection, informed consent, and the possibility of algorithmic bias must also be properly handled. In order to create standards and frameworks for the secure and efficient application of AI in personalized medicine, physicians, data scientists, and regulatory agencies must work together across disciplinary boundaries. By using multidimensional data to forecast therapy responses, optimize treatment regimens, and improve patient outcomes, artificial intelligence (AI) holds the potential to completely transform individualized treatment approaches for autoimmune disorders. AI has the potential to usher in a new era of precision medicine in autoimmune disorders by tackling the difficulties involved in its integration into clinical practice. This would provide patients all around the world hope for more tailored and successful treatments.[48], [49]
Challenges and ethical considerations
While using artificial intelligence (AI) to diagnose and cure autoimmune illnesses has the potential to be revolutionary, there are many obstacles and moral dilemmas to be resolved. Accurate diagnosis and successful treatment of autoimmune disorders are challenging due to their complicated, multifaceted nature, which frequently involves a combination of immunological, environmental, and genetic components. Artificial intelligence (AI) technology, especially machine learning (ML) and deep learning (DL) algorithms, hold promise for improving diagnostic accuracy, finding new biomarkers, and customizing treatment plans. Nevertheless, there are significant obstacles to the use of AI in therapeutic contexts. These include ethical conundrums with patient privacy and permission, technical difficulties with data quality and algorithmic performance, and wider societal ramifications with relation to the fair distribution of AI-enabled healthcare.[50], [51] The completeness and quality of data is one of the biggest obstacles to using AI to treat autoimmune illnesses. Large datasets are essential for AI algorithms to learn and generate precise predictions. However, because different healthcare systems have different diagnostic standards, different recording methods, and different populations, data on autoimmune illnesses can be inconsistent, fragmented, and biased. This lack of consistency may result in AI models that don't fully reflect the range of autoimmune disease presentations or generalize well to a variety of patient populations. Furthermore, the underrepresentation of autoimmune disorders in biomedical databases increases the possibility of biases that could distort AI predictions and perpetuate already-existing inequities in healthcare outcomes and access. [52], [53], [54]
There are also many difficulties with how well AI algorithms work in therapeutic settings. Even though AI models perform exceptionally well in controlled research settings, their practical use frequently shows drawbacks, such as decreased accuracy when confronted to novel or uncommon illness presentations that were not covered in the training datasets. In autoimmune disorders, where patient symptoms can vary greatly and change over time, this problem is especially relevant. Additionally, "black box" AI models pose a concern because their decision-making procedures are opaque to patients or clinicians, making it challenging to understand AI-generated recommendations and thus jeopardizing clinical judgment. It is still quite difficult to make sure AI technologies are visible, interpretable, and fit in with clinical workflows. When using AI in healthcare, ethical issues are crucial, particularly in delicate fields like autoimmune illnesses. Large amounts of personal health data, such as genetic information and comprehensive medical histories, must be handled in order to construct and train AI models, raising privacy concerns.[55], [56], [57] Important ethical requirements include protecting data security and getting informed consent before using such private information. Patients need to understand how their information will be used, the possible hazards, and the privacy protections in place. Furthermore, if the underlying training data reflects historical injustices or does not adequately represent minority groups, AI systems may unintentionally reinforce preexisting biases in the healthcare industry. This could further entrench health disparities by resulting in unequal access to AI-driven diagnosis and treatment tools.[58]
Autonomy and trust are also important factors in the ethical application of AI in the treatment of autoimmune diseases. AI's ability to make or recommend medical decisions may be seen as compromising patient and healthcare practitioner autonomy. To preserve trust in the doctor-patient relationship, it is imperative to make sure AI complements human decision-making rather than replaces it. Additionally, as AI is incorporated into healthcare more and more, physicians need to have the skills and knowledge needed to evaluate AI outputs critically and decide how best to use them in patient care. When it comes to the application of AI in autoimmune illnesses, regulatory and legal frameworks also provide difficulties and ethical issues. There is uncertainty around the approval, supervision, and liability of AI-based products because current regulatory norms frequently fall behind the quick speed of technical innovation. It is crucial to develop strong regulatory guidelines that guarantee the ethical, safe, and effective application of AI in therapeutic contexts. Furthermore, precise rules are required to define accountability, especially when AI-driven choices have the potential to negatively impact patient outcomes.[59], [60], [61]
Future directions
The use of artificial intelligence (AI) in autoimmune disorders is at the forefront of clinical practice and medical research, offering previously unheard-of possibilities to improve the diagnosis, management, and therapy of these intricate illnesses. Future directions that could greatly improve the efficacy and accessibility of AI-driven healthcare treatments for autoimmune illnesses are starting to emerge as the field develops. These developments cover a wide range of topics, such as the construction of patient-centered AI applications, the integration of multi-omics data, the use of AI for early illness detection and prevention, and the development of increasingly complex algorithms.[62], [63]
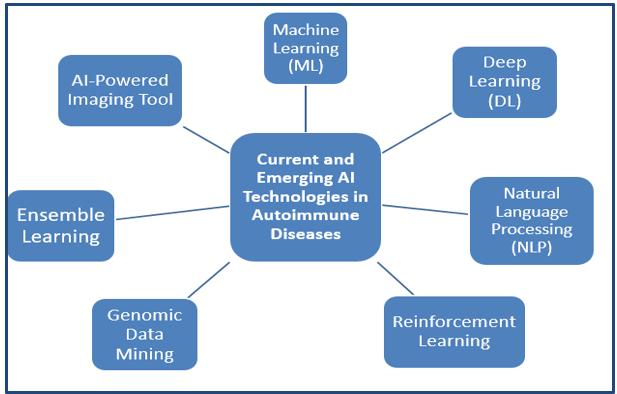
The development of AI algorithms to increase their specificity, sensitivity, and generalizability across a range of patient groups is one exciting avenue for the future. Because they rely on certain datasets that could not adequately represent the variability of autoimmune illnesses, many AI models in this field are currently constrained. Advanced machine learning methods like federated learning, which enables algorithms to learn from a variety of datasets while protecting patient privacy, are probably going to be used in future AI models. By improving diagnostic precision and treatment customization across diverse demographics and clinical contexts, this method can aid in the development of more resilient models that more broadly apply to varied patient groupings. Another crucial area for AI in autoimmune disorders is the integration of multi-omics data, which includes transcriptomics, proteomics, metabolomics, and genomes. AI can provide greater insights into the underlying mechanisms of autoimmune disorders by examining these extensive datasets and finding new biomarkers and pathways implicated in disease pathogenesis. The development of precision medicine tactics may be aided by this integrative approach, enabling more specialized treatments that target the unique biological anomalies causing each patient's illness. Furthermore, by continuously evaluating these intricate data layers in real-time, AI may be able to more precisely forecast disease flares and remissions, facilitating prompt and preventive therapies. The Current and Emerging AI Technologies in Autoimmune Diseases are mentioned in ([Table 4]) with shown in ([Figure 2]).[64], [65], [66].
|
AI Model |
Data Type |
Key Applications |
Advantages |
Limitations |
|
Machine Learning (ML) |
Clinical data, imaging |
Diagnosis, biomarker discovery |
High accuracy with structured data; scalable |
Requires large, high-quality datasets; risk of overfitting |
|
Deep Learning (DL) |
Imaging, genetic data |
Imaging analysis, biomarker discovery |
Superior pattern recognition in complex datasets |
Computationally intensive; "black-box" interpretability |
|
Natural Language Processing (NLP) |
Textual data (EHRs, reports) |
Patient monitoring, data extraction |
Automates analysis of unstructured text; real-time processing |
Limited by language nuances and dataset quality |
|
Reinforcement Learning |
Dynamic patient data |
Treatment optimization |
Learns from feedback; adaptive decision-making |
Requires extensive trial-and-error datasets |
|
Ensemble Models |
Multi-modal data |
Cross-disease diagnosis |
Combines strengths of multiple algorithms |
Complexity in implementation and interpretation |
|
Bayesian Networks |
Genetic, clinical data |
Risk prediction, decision support |
Handles uncertainty well; interpretable outcomes |
Limited scalability with high-dimensional data |
|
Support Vector Machines (SVM) |
Imaging, proteomics |
Classification of disease states |
Effective in smaller datasets; robust classification |
Less effective with noisy, large-scale datasets |
|
Neural Networks |
Imaging, temporal data |
Disease progression monitoring |
Self-improving with more data; handles sequential data |
Prone to overfitting; requires significant computational resources |
|
Disease |
Biomarker Identified |
AI Technique Used |
Clinical Relevance |
|
Rheumatoid Arthritis |
Anti-citrullinated protein antibodies (ACPAs) |
Machine Learning |
Early and accurate diagnosis; predicts disease severity and progression. |
|
Systemic Lupus Erythematosus (SLE) |
Interferon-stimulated genes (ISGs) |
Deep Learning |
Identifies patients with high disease activity; aids in tailoring immunosuppressive therapy. |
|
Multiple Sclerosis |
Neurofilament light chain (NFL) |
Neural Networks |
Tracks axonal damage; useful for monitoring disease progression and therapeutic response. |
|
Psoriasis |
IL-17 and IL-23 cytokine levels |
Support Vector Machines (SVM) |
Differentiates psoriasis from other skin conditions; targets for biologic therapies. |
|
Type 1 Diabetes |
Islet-specific autoantibodies |
Random Forest Classifier |
Enables early prediction of disease onset in at-risk individuals. |
|
Inflammatory Bowel Disease (IBD) |
Calprotectin and lactoferrin levels |
Ensemble Learning Models |
Non-invasive markers for diagnosing and monitoring intestinal inflammation. |
|
Sjögren’s Syndrome |
Salivary gland proteomics |
Principal Component Analysis |
Detects early-stage disease; aids in distinguishing from other autoimmune conditions. |
|
Ankylosing Spondylitis |
HLA-B27 genetic marker |
Genomic Data Mining Algorithms |
Confirms genetic predisposition; supports diagnostic decision-making. |
|
Disease |
AI Tool |
Type of Personalization |
Outcome |
|
Rheumatoid Arthritis |
Machine Learning Models |
Drug dosing optimization |
Improved response to biologics; reduced side effects |
|
Systemic Lupus Erythematosus (SLE) |
Deep Learning Algorithms |
Treatment choice (immunosuppressants) |
Tailored therapy plans based on genetic and immune profiles |
|
Psoriasis |
AI-Powered Decision Support |
Biologic therapy selection |
Enhanced patient-specific treatment; faster remission rates |
|
Type 1 Diabetes |
Reinforcement Learning Models |
Dynamic insulin dosing |
Stabilized blood glucose levels with real-time adjustments |
|
Multiple Sclerosis |
Neural Networks |
Predictive treatment response |
Early identification of non-responders to disease-modifying therapies |
|
Inflammatory Bowel Disease (IBD) |
Ensemble AI Models |
Therapy selection (steroids vs. biologics) |
Reduced time to achieve disease remission and maintenance of symptom-free status |
|
Ankylosing Spondylitis |
Genomic Data Analysis Tools |
Personalized risk-based treatment |
Minimized progression of spinal inflammation through tailored interventions |
|
Sjögren’s Syndrome |
Predictive AI Models |
Drug combination customization |
Reduced symptom severity and improved quality of life |
|
AI Technology |
Current Applications |
Emerging Uses |
Examples in Research or Clinical Trials |
|
Machine Learning (ML) |
Biomarker discovery, predictive modeling for disease flares |
Integrating multi-modal data for holistic disease profiling |
Use of ML to predict lupus flares by analyzing patient records |
|
Deep Learning (DL) |
Analyzing imaging data for early diagnosis |
Automated histopathological grading of tissue samples |
DL algorithms used in rheumatoid arthritis imaging studies |
|
Natural Language Processing (NLP) |
Extracting patient data from electronic health records (EHRs) |
Monitoring patient-reported outcomes in real-time |
NLP-based tools analyzing EHRs for autoimmune disease trends |
|
Reinforcement Learning |
Optimizing treatment regimens based on patient feedback |
Dynamic and adaptive treatment planning |
Reinforcement learning models for drug adjustment in type 1 diabetes |
|
Genomic Data Mining |
Identifying genetic risk factors (e.g., HLA markers) |
Developing polygenic risk scores for personalized medicine |
Studies linking HLA alleles to ankylosing spondylitis risk factors |
|
Ensemble Learning |
Combining multiple algorithms for robust disease classification |
Enhancing cross-disease diagnostic accuracy |
Models combining SVM and Random Forests for autoimmune diseases |
|
AI-Powered Imaging Tools |
Enhancing MRI/CT scan interpretation for inflammatory markers |
Developing portable imaging solutions |
AI-powered tools for detecting synovitis in early arthritis |
|
Wearable AI Devices |
Monitoring real-time symptoms and vitals (e.g., glucose levels) |
Predicting flares via continuous patient monitoring |
Wearable biosensors detecting early inflammatory markers |
Using AI to detect and prevent diseases early is another avenue for the future. The diagnosis of autoimmune illnesses is frequently delayed until substantial tissue damage has taken place, which restricts available treatments and affects patient outcomes. By spotting minute trends in patient data that might point to the early stages of autoimmune illness before clinical symptoms appear, artificial intelligence (AI) has the ability to completely change this paradigm. Utilizing information from wearable technology, electronic health records (EHRs), and patient-reported outcomes, these early detection systems might be incorporated into standard medical procedures to continually monitor patients who are at danger. Early intervention techniques may be made possible by this proactive strategy, which could postpone or even stop the beginning of autoimmune illnesses. The creation of patient-centered AI applications that enable people to take charge of their own health is another aspect of the future of AI in autoimmune disorders. These apps might offer individualized feedback based on each patient's particular medical profile and preferences for medication adherence, lifestyle changes, and disease management. A more involved and knowledgeable patient population might be promoted by AI-driven mobile apps, which, for example, could provide real-time insights and recommendations based on a person's symptoms, activity levels, and biomarker data. Furthermore, by combining complicated medical data into easily comprehensible insights that can be applied to collaborative decision-making, AI may help patients and healthcare professionals communicate more effectively.[67], [68], [69]
One important avenue for the future is the extension of AI's use in clinical trials and research on autoimmune diseases. By more accurately identifying qualified candidates, forecasting patient reactions to experimental treatments, and tracking trial results in real time, artificial intelligence (AI) can simplify the planning and implementation of clinical trials. This could result in the development of new medicines more quickly and affordably, especially for uncommon autoimmune illnesses where small patient populations make traditional trial approaches impracticable. Additionally, AI-driven models can mimic a range of clinical situations, which helps to optimize trial procedures and raises the possibility of positive results.[70], [71], [72]
Notwithstanding these encouraging avenues, in order to optimize its influence, AI in autoimmune disorders must also overcome a number of obstacles. Since the precision and completeness of the data used to train AI models determine their efficacy, maintaining data quality and interoperability is still of utmost importance. Furthermore, to foster confidence and guarantee fair access to AI-driven healthcare solutions, ethical issues pertaining to algorithmic bias, data security, and patient privacy must be strictly controlled. Furthermore, to direct the ethical development and application of AI technologies, interdisciplinary cooperation between physicians, data scientists, regulatory agencies, and patient advocacy organizations will be crucial. AI in autoimmune disorders has the potential to completely transform the way these intricate illnesses are identified, treated, and managed in the future. AI has the ability to greatly enhance patient outcomes and quality of life through increasing algorithmic sophistication, integrating multi-omics data, improving early diagnosis and prevention measures, and creating patient-centered apps. To ensure that AI advancements are both efficient and fair for everyone, it will be necessary to carefully navigate technical, ethical, and regulatory obstacles in order to realize this potential.[73], [74]
Conclusion
Artificial intelligence (AI) has shown great promise in improving disease monitoring, facilitating individualized treatment, and improving diagnostic accuracy in the management of autoimmune disorders. Particular instances highlight the useful applications of AI in clinical contexts, such as the use of deep learning algorithms for imaging-based diagnosis in lupus and machine learning for biomarker discovery in rheumatoid arthritis. Predictive models have also been useful in predicting illness flares, facilitating prompt intervention, and enhancing patient outcomes. Notwithstanding these developments, a number of issues still need to be resolved, such as standardizing data across healthcare systems, guaranteeing AI algorithms' transparency, and handling moral dilemmas pertaining to patient privacy and trust. To create reliable, interpretable AI models that incorporate a variety of data sources, including as genetic, clinical, and lifestyle data, future research should place a high priority on interdisciplinary collaboration.
Source of Funding
None.
Conflict of Interest
None.
References
- CA Janeway, P Travers, M Walport. . Autoimmune responses are directed against self-antigens 2001. [Google Scholar]
- S Kurup, PA Biochemistry. . Biochemistry, Autoimmunity. 2022. [Google Scholar]
- JS Franco, JA Amaya, JM Anaya, JM Anaya, Y Shoenfeld, AR Villarraga. . Thyroid disease and autoimmune diseases 2013. [Google Scholar]
- E Kyritsi, C Magdalini, K Gantenbein. Autoimmune Thyroid Disease in Specific Genetic Syndromes in Childhood and Adolescence. Front Endocrinol 2020. [Google Scholar]
- AP Weetman. Diseases Associated with Thyroid Autoimmunity: Explanations for the Expanding Spectrum. Clin Endocrinol 2011. [Google Scholar]
- MK Desai, RD Brinton. Autoimmune Disease in Women: Endocrine Transition and Risk Across the Lifespan. Front Endocrinol (Lausanne) 2019. [Google Scholar]
- VL Kronzer, SL Bridges, JM Davis. Why women have more autoimmune diseases than men: An evolutionary perspective. Evol Appl 2020. [Google Scholar]
- J Castiblanco, MA Burgos, JM Anaya, JM Anaya, Y Shoenfeld, AR Villarraga. Autoimmunity: From Bench to Bedside. Introduction to Genetics of Autoimmune Diseases 2013. [Google Scholar]
- F Yasmeen, RH Pirzada, B Ahmad, B Choi, S Choi. Understanding Autoimmunity: Mechanisms, Predisposing Factors, and Cytokine Therapies. Int J Mol Sci 2024. [Google Scholar]
- H Touil, K Mounts, PL Jager. Differential impact of environmental factors on systemic and localized autoimmunity. Front Immunol 2023. [Google Scholar]
- R Mazzone, C Zwergel, M Artico, S Taurone, M Ralli, A Greco. The emerging role of epigenetics in human autoimmune disorders. Clin Epigenetics 2019. [Google Scholar]
- A Lossius, JN Johansen, O Torkildsen, F Vartdal, T Holmøy. Epstein-Barr virus in systemic lupus erythematosus, rheumatoid arthritis and multiple sclerosis-association and causation. Viruses 2012. [Google Scholar]
- M T Arango, Y Shoenfeld, R Cervera. . Autoimmunity: From Bench to Bedside 2013. [Google Scholar]
- M Rojas, PR Jiménez, DM Monsalve, Y Pacheco, YA Ampudia, CR Santana. Molecular mimicry and autoimmunity. J Autoimmunity 2018. [Google Scholar]
- NR Jog, JA James. Epstein Barr virus and autoimmune responses in systemic lupus erythematosus. Front Immunol 2020. [Google Scholar]
- P Li, Y Zheng, X Chen. Drugs for Autoimmune Inflammatory Diseases: From Small Molecule Compounds to Anti-TNF Biologics. Front Pharmacol 2017. [Google Scholar]
- LD Quan, GM Thiele, J Tian, D Wang. The Development of Novel Therapies for Rheumatoid Arthritis. Expert Opin Ther Pat 2008. [Google Scholar]
- Q Guo, Y Wang, D Xu, J Nossent, NJ Pavlos, J Xu. Rheumatoid arthritis: pathological mechanisms and modern pharmacologic therapies. Bone Res 2018. [Google Scholar]
- MG Danieli, S Brunetto, L Gammeri, D Palmeri, I Claudi, Y Shoenfeld. Machine learning application in autoimmune diseases: State of art and future prospectives. Autoim Rev 2024. [Google Scholar]
- IS Stafford, M Kellermann, E Mossotto, RM Beattie, BD Macarthur, S Ennis. A systematic review of the applications of artificial intelligence and machine learning in autoimmune diseases. Npj Digit Med 2020. [Google Scholar]
- A Gerussi, M Scaravaglio, L Cristoferi. Artificial intelligence for precision medicine in autoimmune liver disease. Front Immunol 2022. [Google Scholar]
- P Moingeon. Artificial intelligence-driven drug development against autoimmune diseases. Trends Pharmacol Sci 2023. [Google Scholar]
- MI Ahmed, B Spooner, J Isherwood, M Lane, E Orrock, A Dennison. A Systematic Review of the Barriers to the Implementation of Artificial Intelligence in Healthcare. Cureus 2023. [Google Scholar]
- A Visibelli, B Roncaglia, O Spiga, A Santucci. The Impact of Artificial Intelligence in the Odyssey of Rare Diseases. Biomedicines 2023. [Google Scholar]
- H Nosrati, M Nosrati. Artificial Intelligence in Regenerative Medicine: Applications and Implications. Biomimetics (Basel) 2023. [Google Scholar]
- S Capponi, S Wang. AI in cellular engineering and reprogramming. Biophysic J 2024. [Google Scholar]
- H Breiteneder, YQ Peng, I Agache. Biomarkers for diagnosis and prediction of therapy responses in allergic diseases and asthma. Allergy 2020. [Google Scholar]
- A Teniou, A Rhouati, JL Marty. Recent Advances in Biosensors for Diagnosis of Autoimmune Diseases. Sensors 2024. [Google Scholar]
- SS Arya, SB Dias, HF Jelinek, LJ Hadjileontiadis, AM Pappa. The convergence of traditional and digital biomarkers through AI-assisted biosensing: A new era in translational diagnostics?. Biosensors Bioelectronics 2023. [Google Scholar]
- S Shajari, K Kuruvinashetti, A Komeili, U Sundararaj. The Emergence of AI-Based Wearable Sensors for Digital Health Technology: A Review. . Sensors (Basel) 2023. [Google Scholar]
- P Galozzi, D Basso, M Plebani, A Padoan. Artificial intelligence and laboratory data in rheumatic diseases. Int J Clin Chem 2023. [Google Scholar]
- Y Kumar, A Koul, R Singla, MF Ijaz. Artificial intelligence in disease diagnosis: a systematic literature review, synthesizing framework and future research agenda. J Ambient Intell Humaniz Comput 2023. [Google Scholar]
- JD Huang, J Wang, E Ramsey, G Leavey, Tja Chico, J Condell. Applying Artificial Intelligence to Wearable Sensor Data to Diagnose and Predict Cardiovascular Disease: A Review. Sensors 1920. [Google Scholar]
- A Gerussi, M Scaravaglio, L Cristoferi, D Verda, C Milani, E De Bernardi. Artificial intelligence for precision medicine in autoimmune liver disease. Front Immunol 2022. [Google Scholar]
- RW Pettit, R Fullem, C Cheng, CI Amos. Artificial intelligence, machine learning, and deep learning for clinical outcome prediction. Emerg Top Life Sci 2021. [Google Scholar]
- M Bekbolatova, J Mayer, CW Ong, M Toma. Transformative Potential of AI in Healthcare: Definitions, Applications, and Navigating the Ethical Landscape and Public Perspectives. Healthcare (Basel) 2024. [Google Scholar]
- F Ghobadinezhad, N Ebrahimi, F Mozaffari. The emerging role of regulatory cell-based therapy in autoimmune disease. Front Immunol 2022. [Google Scholar]
- MD Rosenblum, IK Gratz, JS Paw, AK Abbas. Treating human autoimmunity: current practice and future prospects. Sci Transl Med 2012. [Google Scholar]
- F Yasmeen, RH Pirzada, B Ahmad, B Choi, S Choi. Understanding Autoimmunity: Mechanisms, Predisposing Factors, and Cytokine Therapies. Int J Mol Sci 2024. [Google Scholar]
- R Farzan. Artificial intelligence in Immuno-genetics. Bioinformation 2024. [Google Scholar]
- S Momtazmanesh, A Nowroozi, N Rezaei. Artificial Intelligence in Rheumatoid Arthritis: Current Status and Future Perspectives: A State-of-the-Art Review. Rheumatol Ther 2022. [Google Scholar]
- EA Poweleit, AA Vinks, T Mizuno. Artificial Intelligence and Machine Learning Approaches to Facilitate Therapeutic Drug Management and Model-Informed Precision Dosing. Ther Drug Monit 2023. [Google Scholar]
- A Mukhopadhyay, J Sumner, LH Ling. Personalised Dosing Using the CURATE.AI Algorithm: Protocol for a Feasibility Study in Patients with Hypertension and Type II Diabetes Mellitus. Int J Environ Res Public Health 2022. [Google Scholar]
- DR Serrano, FC Luciano, BJ Anaya, B Ongoren, A Kara, G Molina. Artificial Intelligence (AI) Applications in Drug Discovery and Drug Delivery: Revolutionizing Personalized Medicine. Pharmaceutics 2024. [Google Scholar]
- J Ye, D Woods, N Jordan, J Starren. The role of artificial intelligence for the application of integrating electronic health records and patient-generated data in clinical decision support. AMIA Jt Summits Transl Sci Proc 2024. [Google Scholar]
- Al Kuwaiti, A Nazer, K Al-Reedy. A Review of the Role of Artificial Intelligence in Healthcare. J Pers Med 2023. [Google Scholar]
- M Alshamrani. IoT and artificial intelligence implementations for remote healthcare monitoring systems: A survey. J King Saud Univ Comp Inf Sci 2022. [Google Scholar]
- SM Varnosfaderani, M Forouzanfar. The Role of AI in Hospitals and Clinics: Transforming Healthcare in the 21st Century. Bioengineering (Basel) 2024. [Google Scholar]
- C Carini, AA Seyhan. Tribulations and future opportunities for artificial intelligence in precision medicine. J Transl Med 2024. [Google Scholar]
- W Tafat, M Budka, D Mcdonald, Mbe, Dr, T W Wainwright. Artificial intelligence in orthopaedic surgery: A comprehensive review of current innovations and future directions. Comp Struct Biotech Rep 2024. [Google Scholar]
- M Afzal, AK Sah, S Agarwal, A Tanzeel, RH Elshaikh, FA Alobeidli. Advancements in the treatment of autoimmune diseases: Integrating artificial intelligence for personalized medicine. Trends Immunother 2024. [Google Scholar]
- J Bajwa, U Munir, A Nori, B Williams. Artificial intelligence in healthcare: transforming the practice of medicine. Future Healthc J 2021. [Google Scholar]
- A Choudhury, O Asan. Role of Artificial Intelligence in Patient Safety Outcomes: Systematic Literature Review. JMIR Med Inform 2020. [Google Scholar]
- M Varnosfaderani, S Forouzanfar. The Role of AI in Hospitals and Clinics: Transforming Healthcare in the 21st Century. Bioengineering 2024. [Google Scholar]
- D Paul, G Sanap, S Shenoy, D Kalyane, K Kalia, RK Tekade. Artificial intelligence in drug discovery and development. Drug Discov Today 2021. [Google Scholar]
- B Khan, H Fatima, A Qureshi. Drawbacks of Artificial Intelligence and Their Potential Solutions in the Healthcare Sector. Biomed Mater Devices 2023. [Google Scholar]
- S Yadav, A Singh, R Singhal, JP Yadav. Revolutionizing drug discovery: The impact of artificial intelligence on advancements in pharmacology and the pharmaceutical industry. Intellig Pharm 2024. [Google Scholar]
- SJ Nass, LA Levit, LO Gostin. The Value and Importance of Health Information Privacy. Beyond the HIPAA Privacy Rule: Enhancing Privacy, Improving Health Through Research 2009. [Google Scholar]
- T Alanzi, A Alhajri, S Almulhim. Artificial Intelligence and Patient Autonomy in Obesity Treatment Decisions: An Empirical Study of the Challenges. Cureus 2023. [Google Scholar]
- J Zhang, ZM Zhang. Ethics and governance of trustworthy medical artificial intelligence. BMC Med Inform Decis Mak 2023. [Google Scholar] [Crossref]
- H Siala, Y Wang. SHIFTing artificial intelligence to be responsible in healthcare: A systematic review. Soc Sci Med 1982. [Google Scholar]
- Li Pomi, F Papa, V Borgia, F Vaccaro, M Pioggia, G Gangemi, S. Artificial Intelligence: A Snapshot of Its Application in Chronic Inflammatory and Autoimmune Skin Diseases. Life (Basel) 2024. [Google Scholar] [Crossref]
- S Momtazmanesh, A Nowroozi, N Rezaei. Artificial intelligence in rheumatoid arthritis: Current status and future perspectives: A state-of-the-art review. Rheumatology and Therapy 2022. [Google Scholar]
- CS Stafie, IG Sufaru, CM Ghiciuc. Exploring the Intersection of Artificial Intelligence and Clinical Healthcare: A Multidisciplinary Review.. Diagnostics (Basel) 1995. [Google Scholar]
- Z Li, S Yin, S Wang, Y Wang, W Qiang, J Jiang. Transformative applications of oculomics-based AI approaches in the management of systemic diseases: A systematic review. J Adv Res 2024. [Google Scholar]
- SA Alowais, SS Alghamdi, N Alsuhebany, T Alqahtani, AI Alshaya, SN Almohareb. Revolutionizing healthcare: the role of artificial intelligence in clinical practice. BMC Med Educ 2023. [Google Scholar]
- IS Forrest, BO Petrazzini, Á Duffy. A machine learning model identifies patients in need of autoimmune disease testing using electronic health records. Nat Commun 2023. [Google Scholar]
- Y Yang, Y Liu, Y Chen, D Luo, K Xu, L Zhang. Artificial intelligence for predicting treatment responses in autoimmune rheumatic diseases: advancements, challenges, and future perspectives. Front Immunol 2024. [Google Scholar]
- J Guo, X Huang, L Dou, M Yan, T Shen, W Tang. Aging and aging-related diseases: from molecular mechanisms to interventions and treatments. Signal Transd Targeted Ther 2022. [Google Scholar]
- C Irissarry, TB Helmchen. Using Artificial Intelligence to Advance the Research and Development of Orphan Drugs. Businesses 2024. [Google Scholar]
- H Chopra. Revolutionizing clinical trials: the role of AI in accelerating medical breakthroughs. Int J Surg 2023. [Google Scholar]
- A U Rehman, M Li, B Wu, Y Ali, S Rasheed, S Shaheen. Role of artificial intelligence in revolutionizing drug discovery. Fundamen Res 2024. [Google Scholar]
- WR Carabali, CC González, LG Sinisterra, LY Heng, E Tsui, S Gangaputra. Managing a patient with uveitis in the era of artificial intelligence: Current approaches, emerging trends, and future perspectives. Asia-Pacific J Ophthalmol 2024. [Google Scholar]
- R Shinde, S Patil, K Kotecha, V Potdar, G Selvachandran, A Abraham. Securing AI-based healthcare systems using blockchain technology: A state-of-the-art systematic literature review and future research directions. Transa Emer Telecommun Technol 2023. [Google Scholar]
- Introduction
- Overview and Classification of Autoimmune Diseases
- Epidemiology and Burden of Autoimmune Diseases
- Pathogenesis of Autoimmune Diseases
- Discussion
- The Role of Artificial Intelligence in Autoimmune Diseases
- Ai in diagnosis and biomarker discovery
- Personalized treatment approaches
- Challenges and ethical considerations
- Future directions
- Conclusion
- Source of Funding
- Conflict of Interest
